NEWS

THE SURFACE FINISH: ESSENTIAL BASIS FOR FOOD SAFETY
|
Stainless steels are iron, chromium and carbon-based alloys with additions of other elements such as mainly nickel (Ni), molybdenum (Mo), manganese (Mn), silicon (Si), titanium (Ti). These alloys are particularly resistant to corro-sion in oxidizing environment for the ability to passivate through adsorption of oxygen. This is due to an amount of chromium (Cr) in the composition of the alloy which must be at least 10.5% with maximum 1.2% carbon, in accordance with EN 10020. In addition to the chromium content, another important prere-quisite for the formation of the passivation film, is the presence of an oxidizing environment (such as the air or water) which promotes its spontaneous process of formation or restoration, in case it gets damaged. The passivation film is fundamental for a good hold of the steel over time, as well as to adequately counteract the various cases of corrosion.In fact, it is necessary to allow the material, both during processing and during use, to be able to exchange with his surroundings a sufficient oxygen quantity, so that it can be considered in the optimal conditions of passivation. Of course, this passivation film can be more or less resistant and more or less anchored to the material depending on the concentration of chromium present in the alloy and depending on the possible presence of other elements (e.g. molybdenum). It is therefore clear that there are different degrees of stainless and corrosion resistance.
|
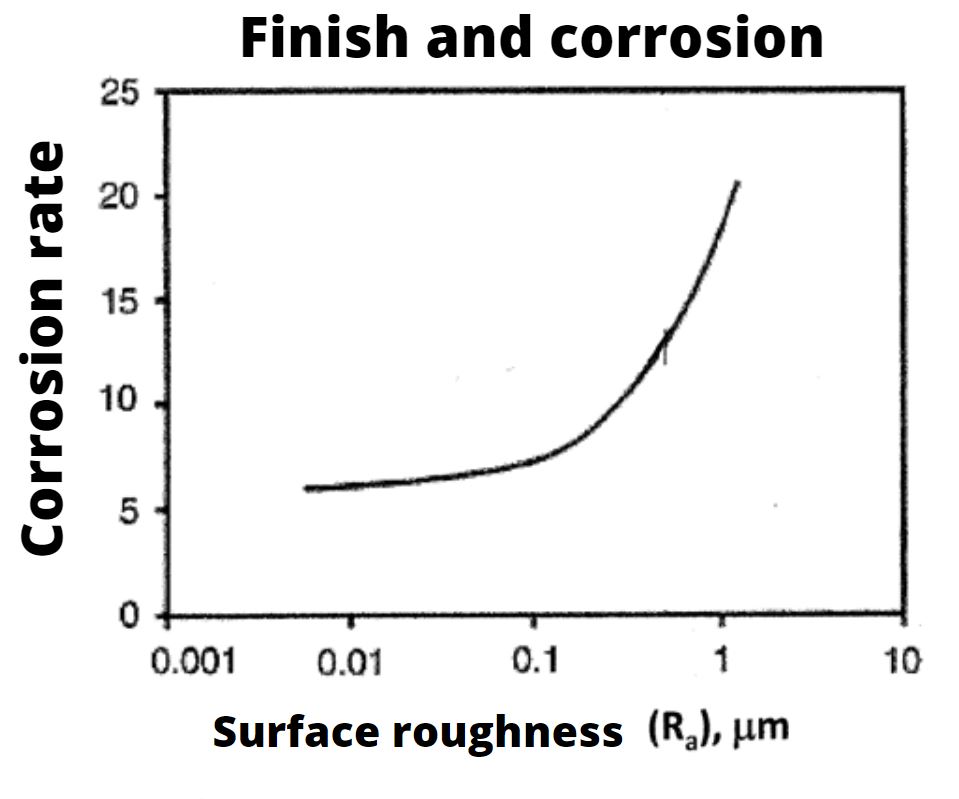
We can consider stainless steel characterized by a good surface finishing in term of low material roughness, a steel that takes good resistance to corrosion. The surface finishing have not only an aesthetic value but, closely related to their roughness, they have a fundamental role in corrosion resistance. For what it con-cerns the stainless steel, the surface finishing can significantly affect the effectiveness of the thin layers of oxides, created by oxidizing environments, determining a peculiar propensity to self-protection with regards to the corrosive agents. Especially for the food industry, stainless steel must have a surface finish that allows excellent cleanability (high bacterial removability and low retentivity). We enclose a graph showing how the surface finish (measured in terms of roughness) significantly affects corrosion; the lower the surface roughness, the lower the corrosion risk will be.